Chapter 1
The Biography of a Trafficking Material
1In 1904, an article titled "Radium and Radioactivity" appeared in Century Magazine, a monthly popular magazine published from 1881 to 1930. The article presents Marie Curie's personal account of the discovery of radium and radioactivity. In the article, Curie discusses in depth her arduous attempts to study the radiation of the compounds of uranium and that of known chemical elements, hoping to discover more which are endowed with atomic radioactivity. She revels that it was the chemists who supplied her with the materials she needed. "As I desired to make a very thorough investigation, I had resource to different chemists, who put at my disposal specimens—in some cases the only ones in existence—containing very rare elements."
2Her next step was to examine different minerals, especially the oxide of uranium ore (pitchblende). To her great surprise, this specimen was found to be four times more active than oxide of uranium itself. The explanation was more than obvious. "The ore must contain a substance more radioactive than uranium and thorium, and this substance must necessarily be a chemical element as yet unknown."1 Her attempts to isolate the new element would lead Marie and Pierre Curie, who joined her research shortly after, to the discovery of both polonium and radium, to the Nobel Prize in Physics in 1903, and to a second Nobel Prize in 1911, this time in chemistry.
3This chapter attempts to present a cultural biography of radium. It is the biography of a material that, from the very first moment of its birth, became involved in the physical system of alpha, beta, and gamma rays and the atomic structure; in the chemical system of atomic weights, emanations, and transmutations; in the medical system of cancer treatments and radon spas; in the commercial system of luminous watches, women's cosmetics, and medical remedies; in the artistic system of luminous paintings and middle-class American culture; and in the industrial system of radium extractions, the production of luminous paint, and the beauty industry.
4During the early twentieth century, radium evolved into a material of the everyday world as well as a "new research tool," in Pierre Curie's terms, of the laboratory site, bringing the above systems into close contact.2 It traveled from laboratories to sites of medical practice and from educational amphitheaters to the physicists' and chemists' workbenches as scientific objects often do. Yet it was also an item of commodification that was transferred from mines and sites of industrial production to those of consumption.
5Anthropologists have long ago argued that in order to understand the concrete, historical circulation of things one has to follow the things themselves "for their meanings are inscribed in their forms, their uses, their trajectories."3 Besides understanding the circulation of the element that is called radium, I want to understand the culture that surrounded its circulation, the transformations in gender relationships that it motivated, the kind of exchanges that took place, and the forms of learned knowledge and gendered skills involved in the circulation of the material. Following around radium, one discovers what historians of science are less familiar with. In addition to being a scientific object with "wonderful and fabulous qualities,"4 radium has been a highly valued commodity. In 1904, its price was $10–$15 per mg and just before the First World War, it escaladed to the astronomical price of $180.5 Radium showed up as a consumer commodity in luminous watches; in women's lotions and creams; in toothpastes, cigarettes, and radium condoms; in ointments for medical use; and in food, drinks, clothing, and endless medical products. Its commodification involved complex political maneuverings, imperial commerce and colonial trade, efforts for monopolization, transformations of women's work, as well as rivalry between scientists and quack doctors over radium quantities, and the exchange of knowledge, skills, and radioactive materials among physicists, chemists, and medical practitioners.
6To come to grips with radium's multiple identities as both a scientific object and a commodity and to explore the paths of its circulation, I developed the concept of a trafficking material. These are materials, which travel from hand to hand, from discipline to discipline, from laboratory to laboratory, or from the scientific world to the world of commodification and consumption. Their main characteristic is their ability to take on multiple identities, not because they are shared between different worlds but because they are transferred across them.6 Motion becomes the inseparable part of their identity which has never been fixed in the first place. Physicists and chemists needed more than a decade after radium's discovery to fully identify the new element, ascribe to and understand its properties, and describe and classify them.7
7Used in scientific laboratories, trafficking materials shift focus from instruments and experimental practices to material substances set on laboratory tables and manipulated by skilled experimenters who aim to reveal the "order of nature" in the distinct cultural setting of the laboratory.8 How do these materials arrive in the laboratory? Trafficking materials such as radium are traded, produced, prepared, and sold, and thus provide a glimpse of how the laboratory and its experimenters are connected to the rest of the mundane world. These materials, furthermore, provide a vital link between the laboratory and the warehouse, the hospital and the academic institution, between sites of production and those of consumption. They are the objects of overlapping networks of knowledge built by different actors. Becoming part of this network it is not only a matter of gaining scientific expertise but also of using the right strategy and of possessing the power to impose oneself. For instance, trafficking materials worked as Trojan horses for sustaining women's experimental work in the field of radioactivity in the early twentieth century. Being able to prepare radium sources for medical use, counsel radiologists about radium's curative properties or perform radium measurements on ocean sediments, women physicists and chemists were able to cross the boundaries of their discipline and move to the fields of medicine or oceanography.
8Trafficking materials possess not only a scientific value but also a social, economic, and sometimes a patriotic one, as we will see in the case of the Viennese physicists of the early twentieth century. They are intimately connected with the knowledge of those who sell them and of those who buy them. They take many journeys and sojourns from production to consumption and scientific exploitation, and they have been used as instruments in order to obtain something else or to restrict and control the kinds of exchanges they are involved in. Moreover, trafficking materials function as devices for reproducing certain relations, often gendered ones, between people in the laboratory, in the work place, or in the market. Thus, trafficking materials are not merely objects but are essential signs in systems of signs such as those that we have already referred to in the case of radium. Mapping the geography of radium's use, a highly complex undertaking, leads to a gender system of laboratory work and is key in understanding how the lively system of science works.
The Radium of the Physicists
9Radium was discovered in France at the end of the nineteenth century, but it was strongly linked to Röentgen's discovery of x-rays in his laboratory in Würzburg, Germany. In late 1895, Röntgen announced that the passage of an electric discharge from an induction coil through a partially evacuated glass tube produces what he called x-rays. The emitted radiation was able to penetrate not only the black paper Röntgen used in his original experiment but thick-material objects as well. In January 1896, Henri Poincaré presented Röntgen's discovery to the French Academy of Sciences, posing the question whether any naturally fluorescent or phosphorescent substance could emit penetrating radiation similar to that of x-rays. To Antoine-Henri Becquerel's satisfaction, that proved to be possible in the case of uranium.9
10The same year, Becquerel observed the darkening of a photographic plate in contact with uranium crystals and described the invisible radiation emitted by uranium. In March 1896, during a meeting of the French Academy of Sciences, Becquerel presented his findings, which were then published within ten days. Shortly afterwards, based on intensive experimentation, he concluded that the emissions were not related to visible fluorescence but to a specific property of uranium.10
11At the time, Marie Curie was working on her doctoral thesis under Becquerel's supervision. She conducted her research on the rue Lhomond at the Ecole municipale de physique et de chimie industrielles (EPCI), in a damp storehouse turned into a physics laboratory. Le hanger (the shed), as the laboratory was known, was directed by her husband, Pierre Curie. Although modest and short of apparatus, the lab provided Marie with enough space to perform her experiments. After the discovery of the new radiation, Marie Curie decided to work on Becquerel rays or uranium rays as they were called at the time. Her aim was to investigate the properties of uranium radiation and a necessary first step was to develop an accurate and reliable method of measuring radiation. Pierre and his brother Paul-Jacques Curie, both prominent physicists working on piezoelectricity and physics of crystals, provided the necessary apparatus.11
12A method based on photographic plates was unassailable for demonstrating the existence of the new radiation but insufficient for measuring its intensity. The density of the exposure on the film, however, could be used as a crude measure of the intensity of the radiation, but it was not accurate enough. Curie aimed for something better. "Instead of making these bodies [the compounds of uranium] to act upon photographic plates, I preferred to determine the intensity of their radiation by measuring the conductivity of the air exposed to the action of rays."12 The ionizing property of the radiation had already been used for the case of x-rays to measure their intensity. Despite their wide usage, instruments such as the gold leaf electroscope and the spinthariscope designed by William Crookes were not accurate and precise enough to be employed for the measurement of uranium rays. The need for new instrumentation became more pressing with the discovery of the new radiation. Pierre and Paul-Jacques Curie had already devised an electrometer in the early 1880s based on the piezoelectric effect of quartz crystals. The apparatus measured small quantities of electricity in absolute terms.
13  Pierre and Paul-Jacques Curie's electroscope.
In the light of the new and slowly emerging field of radiation physics, Marie employed the Curie electrometer in her research, equipped it with an ionization chamber, and transformed it into a reliable tool for ionization measurements. Carrying an electrometer from crystal to radiation physics, Marie demonstrated that the intensity of the radiation was proportional to the amount of uranium. The new method proved to be superior to the photographic plate method. As Marie conducted research on a number of other substances, she soon discovered that only thorium possessed properties similar to those of uranium. Her hypothesis was that radiation was an atomic property unrelated to its chemical structure. Additionally, pitchblende, raw ore containing uranium, was more radioactive than the amounts of included uranium could explain. Obviously, she concluded, there had to be a new radioactive element in the pitchblende. Her experiments with synthetic chalcolite supported her hypothesis and prompted Pierre Curie to involve himself fully in the study of radioactive substances.
Pierre and Paul-Jacques Curie's electroscope.
In the light of the new and slowly emerging field of radiation physics, Marie employed the Curie electrometer in her research, equipped it with an ionization chamber, and transformed it into a reliable tool for ionization measurements. Carrying an electrometer from crystal to radiation physics, Marie demonstrated that the intensity of the radiation was proportional to the amount of uranium. The new method proved to be superior to the photographic plate method. As Marie conducted research on a number of other substances, she soon discovered that only thorium possessed properties similar to those of uranium. Her hypothesis was that radiation was an atomic property unrelated to its chemical structure. Additionally, pitchblende, raw ore containing uranium, was more radioactive than the amounts of included uranium could explain. Obviously, she concluded, there had to be a new radioactive element in the pitchblende. Her experiments with synthetic chalcolite supported her hypothesis and prompted Pierre Curie to involve himself fully in the study of radioactive substances.
14Both Marie and Pierre had approached the problem through physics and had been trained and integrated in that culture. Pierre worked on crystal physics and Marie had been working on the magnetic properties of various minerals under the supervision of the physicist Gabriel Lippmann.13 Deeply devoted to research, Pierre concentrated on the study of the physical properties of the radiation while Marie performed the radiochemical analyses. Between the two, Marie crossed the boundary of her discipline by using methods from chemistry to analyze pitchblende samples while Pierre kept his identity as a physicist rigid. As Davis argues, "If he [Pierre] tended to concentrate on the physics aspect of the work of radioactivity and she on chemistry, this would seem to have been a matter of personal preference."14
15I argue that it was more than a simple preference. As Helena Pycior documents, Marie started to work on radioactivity in December 1897 and it was not before late March 1898 that Pierre joined her.15 Forced by the subject of her research, by the time Pierre got involved, Marie had already integrated chemical methods in the study of the new substances. Her experiments with synthetic chalcolite, the study of all chemical compounds of uranium and thorium, and the testing of all known chemical elements and certain minerals all occurred in this early period. Thus, the core of her own research program required her to continue working on the isolation of new elements.
16In July 1898, after using a combination of electrometric methods and chemical analyses, the Curies discovered polonium. Since they were not members of the French Academy of Sciences, they were not allowed to present their results in the weekly meetings of the academicians. It was Becquerel who presented the discovery on their behalf on July 18.16
17The emergence of the Curies' new research challenged the unity of traditional chemistry. French chemists and academicians soon became uncomfortable, feeling that they might lose control over their discipline. To chemists, outsiders like the Curies seemed to be encroaching on their resources and disrupting disciplinary boundaries. None of the first researchers was a chemist. Marie had primarily studied physics, receiving the Diplôme de licence ès sciences physiques (1893), and a year later, a licence in mathematics as well.17 Pierre had received his licence in physics from the Sorbonne in 1877 and in the early 1880s, collaborating with his brother, discovered the phenomenon of piezoelectricity. At the time of their early research on radioactivity both were working in a physics laboratory, training engineers, and "recruiting students from the écoles primaires supérieures."18 Becquerel, who came from a family with a strong tradition in physics, attended the Ecole Polytechnique in 1873. Two years later, he was appointed as a demonstrator at the Polytechnique and then professor of physics in 1895. That year was significant for him because he was also appointed as a professor of physics at the Museum of Natural History, a position already held by two previous generations of Becquerels. In 1889, he was elected to the Academy of Science in recognition of his work in physics. 19
The Chemistry of the Imponderable
18It was probably because of Becquerel's membership in the Academy of Sciences that French chemists did not overreact. They simply insisted that before the new element could be given any official status, it had to be successfully isolated, its atomic weight had to be measured, and its spectroscopic characteristics analyzed.20 The chemist Gustave Bémont, Pierre's close collaborator and director of the chemistry laboratory next door in EPCI, joined the team and the spectroscopist Eugéne Demarçay was also enlisted as a collaborator. Bémont offered his expertise on the tedious chemical analysis. In December 1898, painstaking measurements and studies of the properties of radiation led the Curies to the discovery of a second element, the one they named radium. Becquerel was once again their representative in the academy. Shortly after, a joint publication by the Curies and Bémont appeared in Comptes rendus, announcing the discovery.21
19It is no coincidence that the publication immediately following that one was Demarçay's "Sur le spectre d'une substance radioactive" (On the spectrum of a radioactive substance"), where he analyzed the spectrum of radium.22 Marie coined the term radioactivity to name the research in radiation physics, rejecting the term hyper-phosphorescence used by J. J. Thomson in England as misleading for the nature of the new radiation.23
20A Nobel Prize in Physics, awarded jointly to the Curies and Becquerel in 1903, affirmed the importance of the new emerging field. In his presentation speech, H. Törneblach, the president of the Royal Swedish Academy of Sciences, referring to the discovery of radium and the findings of Ernest Rutherford and William Ramsay on the release of helium by radium, admitted that they were "discoveries that are bound to be of great importance for the physicist and for the chemist alike."24
21In the years that followed radium's discovery, there was a constant interchange of ideas, practices, techniques, and new knowledge between physicists and chemists. The contribution of the chemists proved to be essential in deciphering the mysteries of the new element. The French chemist André Debierne, the chef de travaux at Marie's laboratory after Pierre's death in 1906, suggested a number of chemical techniques and facilitated the work of physicists employed in the lab. Many young researchers flooded Curie's laboratory, working on the chemistry of the new science, while she was busy perfecting her methods of radium extraction and detecting new radioactive elements. From 1907 to 1914, 58 people worked at Curie's institute, most of them foreigners who came to learn at the source.25
22In the Anglo-American scene, the situation was similar to the one in France. As Lawrence Badash describes, because some of the experiments required chemical separations of radioelements, the physicist Ernest Rutherford "secured the services of a young demonstrator in the chemistry department, named Frederick Soddy."26 At the time, Rutherford was still at McGill University in Montreal, Canada, working on the nature of radium emanation. Harriet Brooks, Rutherford's first graduate student and one of the first women in the field, joined their research and in 1900, she successfully identified radium emanation as a radioactive gas with lower atomic weight than radium. The following year, she moved to the Cavendish Laboratory in England to work with J. J. Thomson and later to Curie's institute where she conducted research with André Debierne as a travailleur libre.27
23The traffic at Rutherford's laboratory, however, "was uncertain" as John Heilborn points out and "brought only a half of dozen people into the laboratory during Rutherford's tenure." Among them was a young German chemist, Otto Hahn, who arrived at McGill in 1905 after spending two years working with Sir William Ramsay in England and with an important discovery in his curriculum vitae. Hahn had identified a new radioelement that emitted thorium emanation and which he labeled radiothorium.28 Meanwhile, Soddy had moved to the Chemical Institute of University College in London to join Ramsay's research. Only when Hahn returned to Berlin and was joined by the Austrian physicists Lise Meitner in 1907 did Germany start to be considered one of the most important radioactivity centers in the world.
24The transfer of knowledge and its carriers—the physicists and chemists who conducted pioneering radium research—was taking place along with the transfer of the materials themselves. Before Rutherford moved to McGill, he was a fellow in the Cavendish Laboratory. A day or two before he left England in 1898, he ordered uranium and radium salts to be sent to his new address and he was also among the few lucky scientists to receive radium substances from Marie Curie as a gift.29 Hahn also did not arrive in Montreal empty handed. With Ramsay's consent, Hahn had taken with him the radiothorium he separated from samples of barium chloride and the actinium that he and a collaborator had earlier proved to be identical to the new element that the German chemist Friedrich Giesel had named emanium. Hahn's radiothorium and actinium salts also followed him to Emil Fischer's laboratory in Berlin.30
25Chemical work required large samples of scarce radium and thus materials mattered even more to the chemists than to the physicists. At the time, one of the most important suppliers for small quantities of radium was Friedrich Giesel, manager of the quinine production at the laboratories of the Braunschweig quinine factory Buchler & Co. Giesel used his chemical expertise for the extraction of radium from uranium ore and, starting in 1901, his factory became the first radium supplier. The Austrian Stefan Meyer, the Curies, and even Dmitrii Mendeleev in Russia received their first samples from Giesel.31 Rutherford was also able to order pure radium bromide from Giesel, taking advantage of a 300-dollar endowment offered by Sir Williams MacDonald, a significant patron of McGill University.32
26Badash alone refers to an amazingly rich traffic of materials in 1913 when the concept of isotopy was at stake. The German chemist Kasimir Fajans sent his student Max Lembert from Karlsruhe to Harvard to work at the laboratory of the atomic weight expert Theodore Richards, an authority in the field. Soddy, who was also involved in research on isotopes, used Ramsay as the mediator to approach Richards as well. Lembert and Richards conducted control experiments on the atomic weight of ordinary lead to be compared with radioactive samples. The trafficking of radioactive samples was impressive. Fajans provided lead previously obtained from Giesel in Braunschweig; Richards got samples from Ellen Gleditsch, the Norwegian chemist who was then working with Boltwood; Ramsay sought supplies from the Cornwall pitchblende and the British Radium Corporation, a new company that established a factory for producing radium bromide under Ramsay's own directorship.33
27Despite the fact that some chemists were eager to probe the mysteries of the new radioelements, the more traditional ones saw radioactivity not only as a physicists' intrusion into their field, but more importantly as an attack on the world view and the basic doctrines of their discipline. When in 1896, Mendeleev proposed the periodic table as a pedagogical way to systematize the existing chemical elements, his primary ordering was by atomic weight or, in his terms, the "elemental weights." Each element had a stable, fixed place in the system depending on its mass. Additionally, it was immutable without any possibility of becoming another element and had no substructure. Up to the end of the nineteenth century, Mendeleev's definition of an element prevailed in the chemical community. In his persuasive account of Mendeleev's work, Michael Gording argues that among the three phenomena that brought chemistry under attack, destabilized the entire discipline, and forced Mendeleev to defend his system was radioactivity.34
28A key step that changed the notion of a chemical element and reshaped chemistry was taken in 1902. Studying the nature of radioactive emanations from thorium, Rutherford and Soddy came up with the theory of atomic disintegration. They concluded that a primary radioactive substance undergoes a series of atomic transmutations; that is: radioactive atoms give birth to a series of atoms of smaller and smaller weights, emitting alpha rays. Therefore, by losing the weight of an alpha particle, an atom would necessarily change species; in other words, one element would become another. This meant that Mendeleev's basic notion of the chemical element was under direct attack. In contrast to the traditional view of a fixed element, a stable state in radioelements could be attained only when the final product was not radioactive. As Rutherford and Soddy argued, "radioactivity is at once an atomic phenomenon and accompanied by chemical changes in which new types of matter are produced. These changes must be occurring within the atom, and the radioactive elements must be undergoing spontaneous transformations . . . the changes in question are different in character from any that have been before dealt with in chemistry."35
29The Rutherford-Soddy theory prompted a couple of remarkable reactions. "What right had physicists to tell chemists their atoms could disintegrate!" exclaimed the American chemist Herbert Newby McCoy at the news of the disintegration theory.36 But while McCoy decided to restore the pride of the chemists by showing that there was indeed a definite connection between uranium and radium, with the second being the daughter product of the first in a decay series, Mendeleev sought to dismiss the transmutation theory as similar to the claims alchemists had put forward long ago.
30The fact that radioactivity was the result of a spontaneous transformation of elements was indeed seen to be a new chemical "alchemy." In 1905, Mendeleev was invited to attend the World's Fair in St. Louis, but a cataract surgery prevented his visit. "Precisely there," he explained to Clemens Winkler, "I intended to put forth my opinion about the semi-spiritualist state into which they [radioactivity researchers] are now trying to enmesh our science. It behooves us to stop it while we can still act."37
31On June 6, 1905, when Pierre Curie visited Sweden to deliver the late Nobel award lecture he was unable to present in 1903, he made clear that the "consequences for chemistry of the properties of the radioactive substances are perhaps even more important" than they are for physics. He was not wrong. To Pierre, the disintegration theory was "more far-reaching than the ordinary chemical transformations: that the existence of the atom is even at stake, and that one is in the presence of a transformation of the elements." Marie Curie definitely affirmed radium's impact on chemistry in her own Nobel lecture in 1911. Radium's discovery marked "incontestably" as Curie claimed, a new epoch for chemists and a kind of chemistry that we might well call "the chemistry of the imponderable."38
32Indeed, the establishment of radium's chemical individuality forced the revision of the notion of a chemical element, allowing the possibility for transmutation, for the existence of atomic substructure and for elements' movement in the periodic table since some of them did not possess a fixed and stable position. Besides revising the chemical theories, new experimental practices and techniques were introduced. Since the presence of radium could often not be detected by the balance or by the spectroscope, novel electrometric methods had to be developed.
33The concept of isotopy and the displacement laws put forward by Soddy and Fajans in 1913 completed the disintegration of the traditional notion of an element and of chemistry as well. According to the rule of an alpha emission, a loss of an alpha particle moved a radioactive element two places to the left in the periodic table. The corresponding rule for a beta emission meant a shift to the right in the periodic table; with the loss of a beta particle, an element moves one place to the right. As a result, a box in the periodic table could hold more than one element. This resulted in the fact that the radioelements produced from the emissions had the same chemical properties as their parents, although they possessed different atomic weights. Thus, the most fundamental effect of radioactivity on chemistry was the redrawing of the periodic table in such a way that could fit the radioelements' changing the primary ordering that was now not the atomic weight anymore but the atomic number of the elements.
The Radium Industry in France and the Austrian Involvement
34  The city of St. Joachimsthal From the discovery of polonium in July 1898 to that of radium in December of the same year, it took the Curies a little more than five months to overcome the academicians' concerns about the nature of their discovery. While the French Academy insisted on the isolation and spectroscopic corroboration of polonium, the Curies needed to process enormous amounts of uranium pitchblende in order to extract a few milligrams of polonium. The only available uranium mine was the one in St. Joachimsthal, Bohemia, then part of the Austro-Hungarian Empire. The mines had a monopoly on the uranium used in industry to color the famous Bohemian glass and porcelain.39
The city of St. Joachimsthal From the discovery of polonium in July 1898 to that of radium in December of the same year, it took the Curies a little more than five months to overcome the academicians' concerns about the nature of their discovery. While the French Academy insisted on the isolation and spectroscopic corroboration of polonium, the Curies needed to process enormous amounts of uranium pitchblende in order to extract a few milligrams of polonium. The only available uranium mine was the one in St. Joachimsthal, Bohemia, then part of the Austro-Hungarian Empire. The mines had a monopoly on the uranium used in industry to color the famous Bohemian glass and porcelain.39
35Since the Curies conducted research in their old, miserable laboratory with insufficient funds and apparatus, they had no chance of approaching the mines directly to ask for a donation of pitchblende. To enlist the support of uranium producers, they had to choose an indirect way and approach those who spoke their parlance. The place to turn to was more or less evident: Pierre and Marie wrote directly to the Kaiserliche Akademie der Wissenschaften (today, the Austrian Academy of Sciences), asking the Austrian academicians to help them obtain 30–100 kilos of residue from the Bohemian mines. "The purpose of this research is exclusively scientific," they emphasized, and thus the academy "would arrange that the administration of the Joachimsthal facilitate our research."40
36 The Austrians had already acquired a reputation as scrupulous and progressive front-line experimenters since the discovery of x-rays. Immediately after Röntgen observed the effect of the new rays and presented his findings to the Würzburg Physical-Medical Society, he sent copies of the report, together with x-ray photographs, to several scientists in Europe. Among them was Franz Serafin Exner, a leading Viennese physicist who received the material on January 4, 1896, including nine x-ray pictures. Amazed by the new phenomenon and persuaded by the impact of observing unexpected images revealing inner parts of the human body, Exner immediately reported the discovery to his colleagues and prompted his advisee Eduard Haschek to work on this topic.  A lady's hand is x-rayed. Three days later, he also reported the event to the Chemical-Physical Society and his brother, the physiologist Sigmund Exner, broke the news to the Society of Physicians.41 Ernst Lecher, a young assistant professor from Prague, attended Exner's presentation. Immediately afterwards, Lecher published a report in the Wiener Presse, edited by his father.42 Die Presse and the rest of the Viennese press devoted long articles to the discovery, including x-ray photographs.
A lady's hand is x-rayed. Three days later, he also reported the event to the Chemical-Physical Society and his brother, the physiologist Sigmund Exner, broke the news to the Society of Physicians.41 Ernst Lecher, a young assistant professor from Prague, attended Exner's presentation. Immediately afterwards, Lecher published a report in the Wiener Presse, edited by his father.42 Die Presse and the rest of the Viennese press devoted long articles to the discovery, including x-ray photographs.
37Around the end of 1896, Sigmund Exner, deeply impressed by the use of x-rays in medicine and physiology, presented an apparatus for the localization of infected areas to the Viennese Society of Physicians. Within the next months, the Viennese clinics anticipated the use of x-rays for medical purposes and various physicians began to use them as a diagnostic tool and to treat skin diseases, tumors, and leukemia.43
38The Curies thus believed they had good reason to seek the assistance of their Austrian colleagues in acquiring the pitchblende for their research. They were not wrong. Eduard Suess, the director of the academy, sent Pierre a telegram assuring him that the mines would provide uranium-ore residues for free. In his reply on November 19, 1898, Pierre praised "the liberal spirit of Suess's government." A month later, the Curies discovered radium, and in appreciation of Austrian's help, they sent an enriched radium sample, a valuable gift, to the Vienna Academy.44
39As Marie Curie later admitted "to try to isolate the supposed new element was a great temptation." In their attempt to separate radium from bismuth, both contained in pitchblende, Marie and Pierre found that by crystallizing the chloride of radioactive barium from a solution, they obtained crystals that were more radioactive and richer in radium than the chloride which remained resolved. Repeating the crystallizations several thousands times they were able to obtain pure chloride of radium.
40The Curie method, the so-called fractional crystallization, was very time consuming, and demanded laborious operations and gigantic amounts of pitchblende to finally yield a few decigrams of radium chloride per ton of ore. Such a procedure clearly demanded the facilities of a factory with enough space to deposit the ore, treat it, and purify it. "Fortunately for us," Curie claimed, "the curious properties of these radium-bearing compounds had already attracted general attention and we were assisted in our research." Among those assisting were the Société Central de Produits Chimiques, which undertook on the Curie's behalf the treatment of the St. Joachimsthal pitchblende in exchange of part of the produced radium.45
41Curie's statement supports Xavier Roqué's argument that her involvement with the radium industry was as essential to her career as her scientific pursuits. In 1904, she and Pierre saw the prospect for the financial support of their institute in Emile Armet de Lisle's attempts to market radium salts. The son of a chemical industrialist, Armet de Lisle extended the family's quinine business located in Nogent-sur-Marne outside Paris, by including the manufacture of radium salts. With the Curies' scientific success, he anticipated the opportunity to create a profitable business. He foresaw the possibility of supplying radium compounds to Curie's laboratory as well as the medical potentials of radium.
42Armet's factory, Sels de radium (Radium salts), soon succeeded in establishing a steady production of radium salts which were then tested and certified by Curie. The factory also functioned as a resource for some of those who wished to visit Curie's laboratory but lucked financial support.46 A few years later, in collaboration with the Bank of Radium of Paris, Curie sent Henry Change, an expert in radioactive minerals, to Colorado to purchase the largest carnotite deposit in the United States. After the final negotiations in 1912, Chagneux forwarded to Paris the papers transferring the property to Curie.47 The fact that historians and biographers have failed to take notice of such connections and Curie's attribution of immense value to the radium industry indicates the lack of concern with the material culture of science.48
Trafficking Materials Among Austrian Industrialists, Government Administrations and Science Practitioners
43While French physicists and chemists focused intensely on radium extraction and purification and explored the industrial dimensions of radioactivity research, Austrian scientists did not want to play the role of mere providers of radium for their French colleagues. Their concerns were scientific as well. The physicist Stefan Meyer, a student of Franz Exner, had already expressed a strong interest in probing radium. In the year of its discovery, Meyer acquired a small sample of radium from Giesel in Braunschweig, to attempt to measure the magnetic permeability of radium.
44According to Meyer's assumption, magnetic permeability depended upon the atomic weight of the elements. Combining the skills of a physicist and a chemist, Meyer extended his research to some 200 inorganic compounds of all the known elements, including radium and polonium. He soon teamed up with the physicist Egon von Schweidler to conduct experiments on the magnetic properties of the radiation of radium and polonium. Those experiments led them to the discovery of the magnetic deflection of the "Becquerel rays" which become known as beta rays, emitted by radium. They were also able to distinguish them from those emitted by polonium (alpha rays).49
45In 1901, given the interest of the Viennese physicists to radium research, Suess and other members of the mathematical and natural sciences division (mathematisch-naturwissenschaften Klasse) of the academy were persuaded by Exner to set up a commission for "the intensive study of the radioactive substances."50 The academy agreed with the Ministry of Agriculture to purchase the necessary raw material from the mines in St. Joachimsthal.
46At the time, physicists were housed in a makeshift institute at Türkenstrasse, a street closely located to the University of Vienna. However, the building proved to be inadequate for conducting radium research. The limited equipment was not the only obstacle. Theoretically, out of 23,000 kg residue, only 12 grams of radium could be extracted.51 Such gigantic amounts of pitchblende required a huge amount of space—as the French had already noticed—experienced chemists, and the facilities of an industrial-like laboratory which the small, improvised Physics Institute was not designed for. 52
47It was Karl Auer von Welsbach who eventually offered to the commission the appropriate space in his factory, located in Atzgersdorf near Vienna. Welsbach had studied chemistry and physics at the Technical University of Vienna, worked at the University of Heidelberg as a lecturer on inorganic chemistry, and later served as Assistent at the second Chemistry Institute in Vienna.53 His patent for a new method to produce incandescent mantle out of 99 percent thorium oxide resulted to the commercial production of mantles in 1886. Welsbach directed the production of the impregnated fluid at Vienna's Chemical Institute while the Pintsch Company in Berlin manufactured the burners. In 1887, he acquired the factory Würth & Co. for chemical-pharmaceutical products in Atzgersdorf and moved the entire chain of production there, creating an affluent gas-lamp industry by improving his patent in the early 1890s.
48At that time, Ludwig Haitinger was the gas-lamp industry's manager.54 Haitinger had studied chemistry at the University of Vienna and worked as an organic chemist before he accepted a position at Welsbach's industry in 1887. He had already conducted research on uranium, rare-earth elements, and the technical improvement of the gas lamp at the Chemistry Institute. Although trained as a scientist, Haitinger's role soon shifted to that of the industry's manager. One of his new tasks was to arrange the exportation of Welsbach's gas lamp to the United States. Three years later, he quit his position and returned as an adjunct to the Chemistry Institute. The technician and chemist won over the industrial manager but only until 1892, when Haitinger returned to the factory as director.
49Combining the scientific and industrial cultures with expertise in chemistry, both Welsbach and Haitinger appealed to Exner and the Austrian Academy as the right persons in the perfect position. Welsbach owned a suitable, spacious, industry-like laboratory for the extraction of radium and was an experienced, innovative chemist already familiar with radioactive elements, having worked with thorium. Haitinger carried over his skills as an experimenter from the Chemistry Institute to Welsbach's industry along with a dexterity with financial issues back to the domain of science.
50In 1901, Suess and Lang drafted a letter to Welsbach on behalf of the Austrian Academy. They proposed to involve him in radium research by using his laboratory either under his or Haitinger's directorship. "Such a research," as Suess and Lang explicitly stated, "cannot be carried out by ordinary aid."55 Obviously, the work on radium introduced a unique and novel cooperative tone into experimentation as chemists and physicists were forced to collaborate with industrialists. Shortly after Welsbach's agreement, the academy's Commission for the Investigation of Radioactive Substances was finally formed, chaired by Exner, and Suess, with Boltzmann, Lang, and Welsbach as members.56 The Austrians, like the French, had finally succeeded in assuring radium's traffic between their own science and industry. In 1903, the Austrian Government placed an embargo on the export of uranium and its residues, marking their power in regulating radium supplies and industry worldwide.57
51After ensuring adequate space for the treatment of pitchblende, the next step for the Austrian Academy was to ensure ample amounts of radium for research. On January 15, 1904, Suess and Lang addressed the Ministry of Agriculture, asking for 20,000 kg residue. As they argued, "For many years, no phenomenon has affected the scientific world in such an extraordinary way as the observation of the strange appearances of radium, a substance that is according to phenomena, an inexhaustible source of light and heat, and thus seems to contradict fundamental assumptions of today's physics." The same day, Suess and Lang emphasized the importance of radium research to the Ministry of Culture and Education. To strengthen their argument, they focused on the Nobel Prize awarded to the Curies for their discovery of radium and the crucial role that Austrians played by providing the radioactive material. They did not fail to mention that the French Academy had economically supported the Curies with 155,000 francs.58 Between the lines, Suess and Lang implied their own demand for financial support.
52While they emphasized the need for a "parallel, up-to-date line" of research in Vienna, comparable to that in Paris, Suess and Lang also negotiated low prices for the pitchblende with the Ministry of Agriculture. Both research groups, in Paris and Vienna, needed 4 grams of radium, which meant at least 10,000 kg of residue apiece. Since the annual production of the mines was only 6,000–7,000 kg of residue, it might have been more than two years before such quantities were available. The negotiation was not an easy one. To persuade the government, the Austrian Academy affirmed that the radium would be used for strictly scientific reasons and that the research was innovative and absolutely important.59
53The deal was eventually closed in March 1904. The mines provided the academy with 10,000 kg residue in two parts and received 8,040 kronen in payment. Welsbach's industry was well compensated for the space provided and for the chemical elaboration of the residues. In 1904, Welsbach received 3,064 kronen and after the extraction of the radium, he received an additional 6,121 kronen. Part of the payment came from a donation that Haitinger made to the academy in the form of a prize for scientific research in his father's memory.60
54Working painstakingly for two years, Karl Ulrich and Haitinger extracted 4 grams of radium bromide out of 30 tons of pitchblende by transferring laboratory methods to the factory and extending their practices to industry. At the same time, they shifted the culture of science from an enterprise focused on teaching and modest experimentation to a modern, relatively expensive research practice. Eventually, the extracted radium constituted the main radioactive material used at the Institute for Radium Research. "With it," Meyer later recalled, "had been laid down the basis for systematic research in this field in Austria," and, as Exner acknowledged, it elevated the institute to one of the richest centers in radioactive materials.61
55The Curies, of course, were regular customers for pitchblende. In addition, the Royal Society of London placed a similar request on May 1, 1904. The English were ready to buy the portions of residues that were not set apart for the Curies.62 Three years later, while both William Ramsay in London and Rutherford in Manchester were involved in radioactivity research, the Austrian Academy of Sciences loaned 350 mg of radium bromide to the English scientists after their persistent requests. Highly respected as he was, Meyer played the role of the mediator. The collaboration of Ramsay and Rutherford soon proved to be unfortunate since the first insisted on keeping the radium for one and a half years, making only radon available to the latter. A second loan in 1908 of 170 mg of radium as 300 mg of radium bromide was sent exclusively to Rutherford for his own experiments.63
56While English and French scientists had already embarked fully on radioactivity research, Emil Warburg, president of the Physikalisch-Technische Reichsanstalt, a physics institute in Berlin, was trying to transform his laboratory into a leading scientific center. As David Cahan notes, by the start of Warburg's presidency in 1905 the institute was not able to meet the demands of contemporary science. Radioactivity was a promising field and Warburg knew the place to start. "I now intend to carry out several works on radioactivity," he wrote to Exner in 1909, "and allow me the following question, whether it is possible to let us have a loan of a small quantity of radium bromide for this purpose." As it is documented in one of the academy's reports, the Commission for the Investigation of Radioactive Substances "had already provided radium to a large number of institutes in France and Germany" first for free and then for moderate prices.64
57By the end of the first decade of the twentieth century, radium had become a trafficking material used by both chemists and physicists, threatening the disciplinary unity of both and blurring their boundaries. For each discipline, radium carried a different identity as it did for the various institutes involved in radioactivity research. The discovery of radium provided the Curies with the prestige they both were lacking when they worked at the EPCI. They also acquired new space and apparatus to continue their experiments. Warburg used radium to transform his institute in Germany into a leading scientific center. For the English also, research on radium was a way to express their aim to maintain their leadership in physics. For the Austrians, however, radium meant much more.
58After the painstaking work of separating radium from pitchblende, Ulrich gained the directorship of the St. Joachimsthal mines, acting as the key link between the Bohemian industry and his colleagues in Vienna.65 Auer von Welsbach, with a serious commitment to both science and industry, was challenged by the opportunity to both contribute to fundamental research and seek financial benefits from new industrial achievements. By using radium as a bargaining tool, the ministries of agriculture and education of the dual Austro-Hungarian monarchy located in Vienna exercised their political strength over the other half of the empire and, after 1903, Austrian academicians such as Suess and Exner proved their administrative and scientific talents by serving as mediators among the Austrian ministries in charge, their international colleagues, and the directors of the Bohemian mines.66
59Exner and the Austrian physicists foresaw in radium research the emergence of an exciting scientific field. Thus, to simply administer the radium sources in St. Joachimsthal and play the role of radium merchants was not part of their goal. As Suess and Lang argued, they had "a kind of moral obligation to the whole scientific world" to pursue research on radium.67 Although not explicit, radium actually meant much more to them. As we will see in the next chapter, radium eventually became the vehicle for obtaining a new physics institute appropriate to the prestige of the Viennese physicists.
Radium in the Clinic
60The discovery of radium brought Austrian physicists into a kind of disciplinary interchange that they had not anticipated. Up to the end of the nineteenth century, although physicists and chemists had been offering their knowledge to medical and pharmaceutical students in the big academic lecture halls, their cooperation ended at the doors of the infirmaries and surgical rooms. Physics and chemistry laboratories were widely open to physicians for educational reasons, but the medical clinics and operating rooms were mainly closed to physicists. However, when it came to using radium for therapeutic purposes in important medical centers in Vienna's fin de siècle, the only way to bring it into the medical facilities required opening the doors to physicists and chemists. The introduction of radium into the operational rooms and cancer centers, involved the intrusion of physicists into the realm of medicine and the constant trafficking of radioactive materials between physics laboratories and sites of medical practice.
61Friedrich Giesel, the German industrial chemist, was the first to observe that radium had biological effects as well. By deliberately applying radium barium bromide to his arm, Bequerel confirmed Otto Walkhoff's observation that radium causes skin wounds. Bequerel and Pierre Curie repeated the experiment and immediately published their results. A picture of Curie's injured forearm after his self-exposure to radium appeared in a French newspaper, bringing the news to the general public.68 The above findings intrigued the medical practitioners. Given the effects of radium on human tissues, many physicians became excited about the possibility of using radium in the treatment of cancer. Pierre was the first to substantiate the interrelation of physics to medicine through radium. He offered a tiny amount of the valuable element to Henri Danlos, a dermatologist at the Saint-Louis hospital in Paris, who put it to good use by treating some cases of lupus.69
62  The laboratory of the physician M. Danysz at the Pasteur Institute. The development of radium therapy in France was astonishingly rapid. Without delay, the radium industries financed the creation and operation of three institutes specializing in this activity. In 1906, Armet de Lisle financed the establishment of the Laboratoire biologique du radium in Paris and three of the most important French radiotherapists, Louis Wickham, Jacques Danne, and Henri Dominici, became associated with the lab. The Laboratoire included research labs for pathology, physiology, physics, and a combined clinical therapy and medical research lab. De Lisle supplied radium from his own industrial refinery in Paris and received a portion of the fee charged to each patient.70
The laboratory of the physician M. Danysz at the Pasteur Institute. The development of radium therapy in France was astonishingly rapid. Without delay, the radium industries financed the creation and operation of three institutes specializing in this activity. In 1906, Armet de Lisle financed the establishment of the Laboratoire biologique du radium in Paris and three of the most important French radiotherapists, Louis Wickham, Jacques Danne, and Henri Dominici, became associated with the lab. The Laboratoire included research labs for pathology, physiology, physics, and a combined clinical therapy and medical research lab. De Lisle supplied radium from his own industrial refinery in Paris and received a portion of the fee charged to each patient.70
63In 1912, the physician and industrialist Henri de Rothschild founded a radium therapy department in his hospital, also in Paris. The same year Jacques Danne initiated the development of the Laboratoire d'essais des substances radioactives in Gif, outside Paris. The apogee of the institutionalization of radium therapy was reached in 1914 with the establishment of Curie's Institut du radium which included two laboratories. One laboratory was dedicated to physics research and the other to biological and medical research. The combination of physics and medicine both housed in the Institut du radium, facilitated perfectly the transfer of materials, instruments, experts, and knowledge. At the same time, industry was closely bound to physics, chemistry, and medicine. 71
64 France had been the pioneer in radium therapy, but it was not the only one. In the following years, a considerable number of radium therapy centers were founded throughout Europe. In 1908, the first center for cancer was established in Stockholm at the university clinic of the Karoline Institute, where the gynecologist James Heyman developed his method of radium treatment of cancer of the uterus. On August 1, 1910, the Radiumhemmet (the Radium Home), a makeshift clinic started by two Swedish doctors, was also founded in Stockholm based on a small donation of 40,000 kronor and 120 mg of radium supply. By 1917, the rapid success of the clinic forced the City Council to provide generous funds and a new well-resourced building.72 In 1910, a similar center was established in Heidelberg by the physician Vinzenz Cerny and in New York at the Memorial Hospital.
65In 1911, King Edward VII of England, following the previous examples, sponsored the Radium Institute in London after his own successful treatment for a lesion on his noise. In Manchester, Rutherford's own city of residence, William Milligan an otolaryngologist, established a radium laboratory at the Manchester Royal Infirmary in 1914. Rutherford was asked to offer his advice on radium sources while his laboratory monitored the radium supplies and their quality for the newly established medical facility.73
66At the General Hospital in Vienna, a radium department was set up with half a gram of radium available in 1912. A year later, a radiological institute was founded at the Vienna Lainz hospital where Heyman's methods were soon used for treatments of cancer. In Denmark, the first radium medical center was established in 1913, with King Christian X as its patron. Interestingly, he bought the valuable element using the money dedicated to the purchase of silver wreaths for King's Frederic VIII's recent death.74
67During the 1910s, biologists, physiologists, and clinical practitioners around the world explored the effects of radium on a number of different human tissues, the nervous system, the eye, as well as on plants and animals. Working on radium, Viennese physicians tied their culture as medical practitioners to that of physicists. They were forced to learn the physics of the new element, to study the way radiation is absorbed by the human body and its dosimetry, and to find the appropriate ways to measure it. Sigmund Exner brought radium research to physiology by studying its effects on animal tissues. Alfred Exner and Guido Holzknecht were the first Austrian physicians to probe the pathology of radium dermatitis. Holzknecht was already recognized as a pioneer in diagnostic radiology and, in 1902 he developed the first instrument to measure x-rays. His device, the chromoradiometer, attracted the interest of the international community of physicians and was tried as a dosimetry apparatus for radium as well. In June 1903, Holzknecht joined Gottwald Schwarz, later director of the Röntgen station at Elisabeth-Spital, in a study of the possible atrophy of the optical nerves due to radium. Soon after, their article appeared in the Mitteilungen of the Society of Physicians.75
68 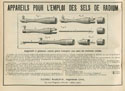 Apparatus designed for the use of radium salts in medicine in 1904.To physicians working on radium therapy, the most astonishing feature of their experimental treatments was their success rate. This success meant that physicists started to dominate the health field. The early methods of radium therapy required the close cooperation of physicists and physicians. The devices were crude and consisted of two main kinds: a) the flat applicators (radium plaques) which were flexible or rigid and designed for external use and b) the spherical ones used in endocavitary applications. They contained a quantity of radium proportionate to the area for treatment. Later on, the enormous cost and lack of radium supplies forced doctors to consider the use of radium emanation instead. Physicists, responding to physicians' needs, designed more sophisticated devices, such as steel needles containing capillary glass tubes filled with radon and treatment was based on needle puncture.76
Apparatus designed for the use of radium salts in medicine in 1904.To physicians working on radium therapy, the most astonishing feature of their experimental treatments was their success rate. This success meant that physicists started to dominate the health field. The early methods of radium therapy required the close cooperation of physicists and physicians. The devices were crude and consisted of two main kinds: a) the flat applicators (radium plaques) which were flexible or rigid and designed for external use and b) the spherical ones used in endocavitary applications. They contained a quantity of radium proportionate to the area for treatment. Later on, the enormous cost and lack of radium supplies forced doctors to consider the use of radium emanation instead. Physicists, responding to physicians' needs, designed more sophisticated devices, such as steel needles containing capillary glass tubes filled with radon and treatment was based on needle puncture.76
69Given the lack of a suitable method for standardizing radium preparations, the expanded use of radium in cancer therapy necessitated closer relationships between physicists and physicians. Physicians were in desperate need of accurate methods for radiation measurement. Their major concern was the precise description that could ultimately lead to successful duplication of their work. It was the physicists who had the expertise and the instrumentation to do so.
70In 1910, during its meeting in Brussels, the Congress of Radiology and Electricity decided to appoint a committee for the preparation of an International Radium Standard. Mme. Curie was asked to prepare the original standard and the Austrians agreed to produce a similar one to be compared for accuracy reasons. Having the monopoly of the Bohemian uranium mines, the Austrian physicists were in charge of radium supplies in Europe and regulated the radium prices. For example, by 1908, the Austrian Academy of Science had already provided radium to a number of hospitals such as the general hospital and university clinics in Vienna and the university clinic in Krakau.77
71However, the traffic of the material was both ways. On the one hand, hospitals' demands for radium increased rapidly with the establishment of radium laboratories within their facilities and thus also increased the dependency of medical practitioners on the physicists. On the other hand, given radium's astronomical price, the physicists were glad to reuse the spent radon needles received from hospitals for their own research. It was becoming apparent that physicists and medical practitioners were more and more in a mutually dependent relationship. It is indicative that in the process of organizing the second Congress on Radiology and Electricity, Rutherford admitted to Meyer that "we have very few medical representatives in the list, and this is invidious." However, on June 29, 1914, while Rutherford was arranging the last details for the meeting, he did not anticipate the outbreak of World War I in August 1914.78
Radium as a Medical Commodity
72  This article advertised radium's power to "bleach the Negro." In early-twentieth-century American society, radium was deployed in an environment of strong racial discrimination. As early as 1904, a chemist at the University of California designed a series of experiments using radium to "bleach the negro" and turn his skin white. Radium combined with x-rays was tested for its effects on the coloring cells of the body. The case made headlines: "burning out birthmarks, blemishes of the skin, and even turning a negro white with the magic rays of radium, the new mystery of science."79 The radium craze combined with the eugenics movement of the 1920s in the U.S. made possible articles dealing with issues such as "sterilization in the interest of race betterment" to sully radiology journals and present radium as a commodity ready to serve the medical world.80
This article advertised radium's power to "bleach the Negro." In early-twentieth-century American society, radium was deployed in an environment of strong racial discrimination. As early as 1904, a chemist at the University of California designed a series of experiments using radium to "bleach the negro" and turn his skin white. Radium combined with x-rays was tested for its effects on the coloring cells of the body. The case made headlines: "burning out birthmarks, blemishes of the skin, and even turning a negro white with the magic rays of radium, the new mystery of science."79 The radium craze combined with the eugenics movement of the 1920s in the U.S. made possible articles dealing with issues such as "sterilization in the interest of race betterment" to sully radiology journals and present radium as a commodity ready to serve the medical world.80
73The link between radium's "magic" power and its commodification as a medical item was reinforced by doctor's public claims of its therapeutic effectiveness. As C. Chase reported, over 80 articles on radium therapy had appeared in the North American literature by 1906. Medical doctors created lengthy lists of conditions that apparently benefited from radium treatment and famous scientists such as Frederick Soddy suggested radium as a great therapeutic agent.81 Worried by physicians' concerns about radium, Rutherford wrote to Stefan Meyer: "I hope you will be able to reserve a large quantity [of radium] for experimental purposes, otherwise I am afraid it will all go into the doctors hands." 82
74The potential of radium in medicine and as a cure for cancer drove doctors to investigate the new element. Exemplary was the suggestion of Sir Alfred Pearce Gould, the famous cancer specialist at Middlesex Hospital in London, to purchase radium as a gilt-edged investment for hospital funds. At a hospital board meeting in November 1913, Gould urged his colleagues to buy radium as there was no more permanent way of investing money for the future. He added that "as to the use of radium I must speak guardedly. It is of the greatest importance in cancer case."83
75  The general laboratory at the Standard Chemical Company in Pittsburgh.Industrialists seemed to have had a similar view about radium's potentiality as a valuable commodity. The Standard Chemical Company, one of the largest U.S. commercial producers of radium, was established in 1911 by Joseph Flannery, a successful Pittsburgh businessman. Although he had no scientific or technical knowledge, Flannery set up a lucrative radium extraction plant in nearby Canonsburg, Pennsylvania. He sought to refine radium from ore mined, sorted, sacked in, and transported from Colorado mines. A barium-radium chloride solution was then transferred to a refinery laboratory in the Oakland section of Pittsburgh. The refinery employed 15 technicians in addition to 150 workers in Canonsburg who performed the first radium process. Bankers' investments, chemical technology, and Flannery's business genius were all decisive. Production relied on around 15 chemists whose laborious work and precision laboratory analysis yielded just one gram of radium every five weeks. Its price, however, was 120,000 dollars, a value at the time greater than the weight-based cost of the Hope diamond.
The general laboratory at the Standard Chemical Company in Pittsburgh.Industrialists seemed to have had a similar view about radium's potentiality as a valuable commodity. The Standard Chemical Company, one of the largest U.S. commercial producers of radium, was established in 1911 by Joseph Flannery, a successful Pittsburgh businessman. Although he had no scientific or technical knowledge, Flannery set up a lucrative radium extraction plant in nearby Canonsburg, Pennsylvania. He sought to refine radium from ore mined, sorted, sacked in, and transported from Colorado mines. A barium-radium chloride solution was then transferred to a refinery laboratory in the Oakland section of Pittsburgh. The refinery employed 15 technicians in addition to 150 workers in Canonsburg who performed the first radium process. Bankers' investments, chemical technology, and Flannery's business genius were all decisive. Production relied on around 15 chemists whose laborious work and precision laboratory analysis yielded just one gram of radium every five weeks. Its price, however, was 120,000 dollars, a value at the time greater than the weight-based cost of the Hope diamond.
76  Containers intended for transporting radium needles or tubes for medical use.As a final commodity radium lived several lives designed by employees who were engaged in sales, clerical, and management activities at the company's offices in Pittsburgh.84 Radium showed up in quite a few medical products designed and promoted by the Standard Chemical Company. Containers for small radium needles made with a mix of nickel, copper, and zinc (but not iron or lead which could have been the safest for transporting radium) were placed in a small velvet-lined leatherette case, carrying the company's stamp. Later, steel cases replaced the older ones and were designed to store radium plaques. Lead-lined boxes containing radium salts, radium needles, radium solutions for drinking and intravenous use, and radium compresses for pain relief proved the "unquestionable value of radium."
Containers intended for transporting radium needles or tubes for medical use.As a final commodity radium lived several lives designed by employees who were engaged in sales, clerical, and management activities at the company's offices in Pittsburgh.84 Radium showed up in quite a few medical products designed and promoted by the Standard Chemical Company. Containers for small radium needles made with a mix of nickel, copper, and zinc (but not iron or lead which could have been the safest for transporting radium) were placed in a small velvet-lined leatherette case, carrying the company's stamp. Later, steel cases replaced the older ones and were designed to store radium plaques. Lead-lined boxes containing radium salts, radium needles, radium solutions for drinking and intravenous use, and radium compresses for pain relief proved the "unquestionable value of radium."  Radium emanator produced by the Standard Chemical Company for treating two people. Standard radium emanators for treating two people and even a portable office-style emanator for treating one person were designed, produced, and sold by the company. The emanator was an unusual device intended to be placed overnight in water so that a ready supply of radioactive water would be available the next day. Its functioning principle was based on the fact that radium emanation or radon when inhaled had therapeutic properties. During the 1920s, several other companies such as the Radium Life, Inc. of Los Angeles, the Radium Company of Colorado, the Radium Chemical Company of New York City, and the Canadian Radium and Uranium Corporation controlled the radium market and supplied hospitals with all kinds of radium applicators, needles, and radon seed implanters and capsules, all medical technologies used in cancer therapy.
Radium emanator produced by the Standard Chemical Company for treating two people. Standard radium emanators for treating two people and even a portable office-style emanator for treating one person were designed, produced, and sold by the company. The emanator was an unusual device intended to be placed overnight in water so that a ready supply of radioactive water would be available the next day. Its functioning principle was based on the fact that radium emanation or radon when inhaled had therapeutic properties. During the 1920s, several other companies such as the Radium Life, Inc. of Los Angeles, the Radium Company of Colorado, the Radium Chemical Company of New York City, and the Canadian Radium and Uranium Corporation controlled the radium market and supplied hospitals with all kinds of radium applicators, needles, and radon seed implanters and capsules, all medical technologies used in cancer therapy.
77The constant trafficking of the valuable commodity among industry, science, and technology was best elaborated in France. Radium was what Marie Curie and the industrial chemist Armet de Lisle certainly saw as the most important new commodity. While Curie used industry to serve her physical and chemical investigations of the new element, de Lisle used science and technology to extend the radium market to medical applications. "Indeed he became one of the most important protagonists of radium therapy in France" writes Soraya Boudia.85 de Lisle's radium factory became the major supplier of medical technologies in France during the 1910s. Several apparatus were advertised through the journal Le Radium, mainly devices for the internal treatment of cancer.
78  A standard radium emanatorium, where men were treated for several kinds of diseases. In Austria, medical consumption of radium was intimately connected to curative baths. In June 1904, Heinrich Mache, Exner's student and Assistent, conducted the first study of the water at the famous health spas in Gastein. Bubbling air through bottles completely filled with the Gastein water, Mache obtained emanation that he soon concluded was due to radium minerals in the earth's crust. Within the next three months, he completed his experiments, giving a much fuller account of the amount of radioactive emanation in the water. Soon thereafter, Meyer joined Mache and they expanded their investigations to other springs such as Carlsbad, Marienbad, Teplitz-Schönau-Dux, Franzensbad, and St. Joachimsthal. Their results varied, but the St. Joachimsthal water proved to contain the largest amount of emanation found in any spring water. Given that the radium content in the ground was high, Mache's earlier conclusion proved to be right.86 The presence of radium emanation, later called radon gas, in the spring water explained its therapeutic properties. In 1904, Mache proposed a measurement unit for the concentration of radon in water that took his name.87 Both the scientific world and the popular culture embraced radioactive emanations as miraculous remedies, gifts to humanity from the foremost geniuses of an inventive age.
A standard radium emanatorium, where men were treated for several kinds of diseases. In Austria, medical consumption of radium was intimately connected to curative baths. In June 1904, Heinrich Mache, Exner's student and Assistent, conducted the first study of the water at the famous health spas in Gastein. Bubbling air through bottles completely filled with the Gastein water, Mache obtained emanation that he soon concluded was due to radium minerals in the earth's crust. Within the next three months, he completed his experiments, giving a much fuller account of the amount of radioactive emanation in the water. Soon thereafter, Meyer joined Mache and they expanded their investigations to other springs such as Carlsbad, Marienbad, Teplitz-Schönau-Dux, Franzensbad, and St. Joachimsthal. Their results varied, but the St. Joachimsthal water proved to contain the largest amount of emanation found in any spring water. Given that the radium content in the ground was high, Mache's earlier conclusion proved to be right.86 The presence of radium emanation, later called radon gas, in the spring water explained its therapeutic properties. In 1904, Mache proposed a measurement unit for the concentration of radon in water that took his name.87 Both the scientific world and the popular culture embraced radioactive emanations as miraculous remedies, gifts to humanity from the foremost geniuses of an inventive age.
79As the New York Times reported in 1911, public opinion forced the government to assume control of the exploitation of these natural sources and establish a curanstalt, a therapeutic institution accessible to all classes and open year-round. The Austro-Hungarian monarchy subsequently asked for the supervision of Mache and Meyer. The curanstalt was operated by pipes drawing the water directly from the mines. The water was filtered without losing anything of its radioactivity and directed to the baths. A luxurious bath was then built and inhalation apparatus were designed. Visitors came from near and far to soak in the supposedly therapeutic water and inhale the air. Patient prescriptions were given in Mache units. As Meyer claimed later, concerning the medical uses of radium, "Austria was in this respect the best country by means of research." 88
80  The advertisement of the Radiumwerk Neulengbach. The interest in radium for medical use was so great that radium preparations and apparatus to produce radioactive water quickly became commercially available in Vienna. For example, the Radiumwerk Neulengbach, a commercial supplier of radium preparations, opened a store near the Physics Institute. They advertised their products as being as radioactive as the natural springs and effective against a long list of medical problems such as neuralgia, neurosis of the digestive organs, and malignant neoplasm.89 Radium had become firmly rooted in everyday human life as anyone could go to the local store to buy a radium emanator, a radium ointment, or a radium compress.
The advertisement of the Radiumwerk Neulengbach. The interest in radium for medical use was so great that radium preparations and apparatus to produce radioactive water quickly became commercially available in Vienna. For example, the Radiumwerk Neulengbach, a commercial supplier of radium preparations, opened a store near the Physics Institute. They advertised their products as being as radioactive as the natural springs and effective against a long list of medical problems such as neuralgia, neurosis of the digestive organs, and malignant neoplasm.89 Radium had become firmly rooted in everyday human life as anyone could go to the local store to buy a radium emanator, a radium ointment, or a radium compress.
Radium as a Commodity
81"When we [Mache and Meyer] visited it [the city of St. Joachimsthal] in 1904, the city was up to the spirit of the time. There was a variety of 'roasted meet with radium,' (Radiumbraten) 'radium soap,' 'radium cigars,' 'radium cigar holders,' 'radium pastries,' etc." So wrote Stefan Meyer, describing his visit to the city of the uranium mines in Bohemia. "One owner of a guesthouse called it Radium-Gaststätte. This irritated the competitors. Hence another one called his own Zur Emanation and he ordered the printer to imprint this name on the labels of cigar holders. He wanted to distribute them as a way of advertising his guesthouse. But the word was strange to the printer and thus he printed the most familiar to him, Zur Imitation."90 Maybe the Bohemian printer was unfamiliar with the word "emanation," but by the mid-1910s, hardly a person either in Europe or in the United Sates was unfamiliar with the word "radium."
82A very rapid expansion in manufacturing and consumer products that contained radium was one of the most dramatic phenomena of the early twentieth century. Radium cleanser, for example, was a sprayer that supposedly killed flies, mosquitoes, and germs as well as cleaned furniture, paintwork, and porcelain. Radium Enamel, manufactured by J. L. Prescott Co. of New York, was a radium product for shining metal surfaces, and Radium was a boot polish made in England by Radium Polishes Ltd. In Germany, chocolate bars containing radium were sold as a "rejuvenator." Radium-brand creamery butter, keyholes and chains, and toys and military equipment coated with radium flooded the market. A trade radium preparation called Nirama was used as a fertilizer to increase the speed of growth in potatoes, flowers, and other plants. The miracle of science was being revealed in mundane things. At the 1904 banquet of the New York Technology Club, well-respected New Yorkers toasted with liquid glowing radium cocktails. Radium roulette, a New York rage, appeared the same year. Participants gambled and played with wheels, balls, and chips all layered with luminescent radium paint.91
83 The radium products that Meyer saw in St. Joachimsthal were another indication of the public's radium obsession that was decisively shaping both the European and American cultures during the 1910s and 1920s. Even the radium pastries in St. Joachimsthal bakeries contained minute quantities of radium. Saved envelopes describing the production of bread with radium water by Hippmann-Blach, a known bakery in the city, attest to the habits of the day.
84 Radium-brand cigars were produced by the A. Batschari tobacco company in Baden, Germany, and later also by the Polus tobacco company in Balerna, Switzerland. Radium was added to toothpastes such as Doramad, a German brand produced by Auergesellschaft in Berlin. Advertising and product labeling persuaded believing consumers. The toothpaste label indicates that "its radioactive rays" strengthen teeth and gums, providing "a new, pleasant, mild, and refreshing taste." In the brochure for the promotion of Doramad, under the title, "What is biologically important?", a joyful woman with a shiny smile stares at her reflection in the mirror.
85  Vita Radium suppositories for men's sexual rejuvenation. Both women and men were the targets of radium advertising but for entirely different products. Men of that era were introduced to radium condoms and were promised a better sexual life. "The best radium finest seamless male pouches," moreover, "guaranteed for one year" appeared in the U.S. around the 1920s, and Radium Nutex, another condom brand came out sometime later. The most impressive product attracting "weak discouraged men" that could now "bubble over with joyous vitality" appeared around the end of the 1920s and was manufactured by the Home Products Company of Denver in Colorado. Vita Radium Suppositories promised to restore men's sex power because, as the advertisement argued, "a man must be in a bad way indeed to sit back and be satisfied without the pleasures that are his birthright." Soluble radium was added to a cocoa butter base in the form of a suppository and was introduced into the rectum in order to stimulate "the weakened organs that needed its [radium's] vitalizing aid.92
Vita Radium suppositories for men's sexual rejuvenation. Both women and men were the targets of radium advertising but for entirely different products. Men of that era were introduced to radium condoms and were promised a better sexual life. "The best radium finest seamless male pouches," moreover, "guaranteed for one year" appeared in the U.S. around the 1920s, and Radium Nutex, another condom brand came out sometime later. The most impressive product attracting "weak discouraged men" that could now "bubble over with joyous vitality" appeared around the end of the 1920s and was manufactured by the Home Products Company of Denver in Colorado. Vita Radium Suppositories promised to restore men's sex power because, as the advertisement argued, "a man must be in a bad way indeed to sit back and be satisfied without the pleasures that are his birthright." Soluble radium was added to a cocoa butter base in the form of a suppository and was introduced into the rectum in order to stimulate "the weakened organs that needed its [radium's] vitalizing aid.92
86In the 1920s, men were also advised to use the Radiendocrinator, which was "the last word in scientific manufacture," as the American Endocrine Laboratories assured the customers of this aphrodisiac product. The 150-dollar gold-plated Radiendocrinator consisted of seven radium-soaked pieces of paper about the size and shape of a credit card, covered with a thin piece of clear plastic and two gold-wire screens placed in a beautiful, dark, embossed leatherette case. It was fashioned by William Bailey, a Boston swindler who promoted his product as appropriate for "sexual rejuvenation." This scam item was supposed to restore and enhance the male sex drive when it was worn as an athletic strap under the scrotum during the night.
87 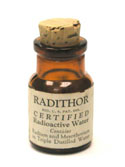 A bottle of Radithor, manufactured by Bailey Radium Laboratories of East Orange, New Jersey. However, Bailey's most amazing product of all was Radithor, a 1920s radium tonic which contained more than 2μCi (74kBq) of radium per bottle. Produced by concentrated radium and mesothorium fluid diluted with distilled water, Radithor was packaged in half-ounce bottles and sold by the case of thirty. Between 1925 and 1930 more than 400,000 bottles were consumed by men who wished to restore their virility. One of the first victims of Radithor was Eben MacBurney Byers, a wealthy steel manufacturer from Pittsburgh and well-known sportsman and playboy who died of radium poisoning in 1932. At the recommendation of his doctor he averaged three bottles of Radithor a day for two years. 93 His case was decisive in the passage of the Food, Drug and Cosmetic Act in the U.S. Congress in 1938 which mandated premarket approval of all new drugs and cosmetics.
A bottle of Radithor, manufactured by Bailey Radium Laboratories of East Orange, New Jersey. However, Bailey's most amazing product of all was Radithor, a 1920s radium tonic which contained more than 2μCi (74kBq) of radium per bottle. Produced by concentrated radium and mesothorium fluid diluted with distilled water, Radithor was packaged in half-ounce bottles and sold by the case of thirty. Between 1925 and 1930 more than 400,000 bottles were consumed by men who wished to restore their virility. One of the first victims of Radithor was Eben MacBurney Byers, a wealthy steel manufacturer from Pittsburgh and well-known sportsman and playboy who died of radium poisoning in 1932. At the recommendation of his doctor he averaged three bottles of Radithor a day for two years. 93 His case was decisive in the passage of the Food, Drug and Cosmetic Act in the U.S. Congress in 1938 which mandated premarket approval of all new drugs and cosmetics.
88As women's sexuality was not yet at the top of any industry's interests, early advertisements that targeted women promoted mainly radium cosmetics. The information that caught the attention of many women as early as 1904 was the claim that "radium and x-rays [can be] used to beautify." Radium was viewed as a godsend panacea that was able to safely remove even birthmarks.94 During the 1920s, beauty was engineered in the intersection of fashion, science, and business. Social trends and "scientific" innovations fueled by appropriate forms of mass advertisement resulted in the production of a series of contraptions and quark medicines that women willingly used to captivate the male eye. Extravagant claims for the aesthetic benefits of radium products linked women's beauty to the uses of radium lipsticks, face creams, and powders. Beauty products such as Radior soaps, face creams, and lotions were guaranteed to contain "a definite amount of Actual Radium—nature's greatest aid to Beauty." Already in 1918, several Radior toilet requisites were produced by Radior Co. Ltd. of London and sold in Fifth Avenue shops in New York. Since there were no Food and Drug Administration (FDA) regulations for premarket approval of new cosmetics at that time, Radior products jumped right into the sales market. Full page advertisements in major New York newspapers argued that, "It is easy to prove the superiority of 'Radior' preparations. Try them. See how smoothly the vanishing cream rubs in." The 5,000-dollar guarantee the company promised to the unsatisfied customer was not enough to relieve women's radiation burns, scarring, and skin cancer that soon appeared.95
89In the early 1930s, this time in France, a Parisian doctor, Alfred Curie, and a pharmacist, Alexis Moussali, epitomized the beauty trend in the industry and launched a cosmetic brand Tho-Radia "for pharmaceutical products, beauty products and perfumes." A publication in La Revue des specialties that appeared immediately after its launch, argued that the cream, with a base of thorium and radium, had great success in Paris with promising curative and beautifying properties. The company's advertisements spoke of a "scientific cream" which was promoted and sold to the entire country thanks to the public enthusiasm. Besides the cream, Parisian women had a variety of choices among powders, lipsticks, and toothpastes, all Tho-Radia products. 96
90While mass advertisement shaped women's consumerism and suggested beauty innovations, new technology created the means. In the late nineteenth century, more than 1,000 patents were used for new corset designs, a technological enhancement that supported women's bust from below. In the early 1930s, the radioactive corset was added to the rubber, electric, and magnetic corsets. It was called the Shilouette Radiante and was manufactured by Shilouette, a German company with subsidiary companies in London and Paris. In 1937, the partner from the Paris office obtained a patent for the new radioactive product and Shilouette launched in London as well. Its immediate success was due to the fact that it had a "stimulating, even rejuvenating influence on the cells of the human body, aids fatigue, warms the body, and helps rheumatic pain." 97
91The combination of scientific authority, mass advertising, and commercialization of women's beauty products enforced the use of radium as a highly valued commodity. Radium dial painters of the 1920s, the women who applied radium paint on watch dials in several factories in the United States, had direct access to the material and grabbed the chance to use the sensational product at no cost. While artificial phosphorescence was a novelty at the time, women applied radium directly to their buttons, fingernails, faces, and eyebrows during their daily shift. "And I can remember, too," revealed one of the dial painters to a reporter of the New Yorker "a lively Italian girl at the plant who painted her teeth with the stuff [radium paint] whenever she had a date, so that they'd shine in the dark."98 The luminous paint, however, sentenced the painters to a slow and painful death as all of them pointed their brushes between their lips to give the brush an exceedingly fine point and speed up their work. Radium dial timepieces, clocks, and especially watches were popular with both the civilian and the military populations, so the women's work was important to both the U.S. army and navy and those corporations involved in the production. While the radium industry was boosted, women's daily radium ingestion resulted in painful deaths and the most infamous case of radium poisoning in the history of industrial occupation accidents. Commerce, war, and the strong belief in science and progress helped turn radium into a valuable thing. Moreover, commodity culture selectively effaced cases of radium beauty horrors and radium poisoning from the mass memory.99
Trafficking Materials, Conflicted Images
92Trafficking materials have the precarious status of having an identity in constant transition. Throughout the early twentieth century, radium embodied several, sometimes conflicted, images to those who produced it, traded it, studied it, and consumed it. In the eyes of the scientists, radium represented a strategy to organize their work place, a means to make a name in the competitive world of science, or a medium with which to reveal a piece of the natural world. To industrialists, radium coincided with the stability and prosperity of their business or was connected to their ambitions of being part of the modern world filled with radium refreshments, aphrodisiac radium products, toiletries, and cigars. To the physicians, radium became inseparable from their identity as progressive practitioners; it forced them to cooperate with physicists and technicians and to trade with industrialists and companies producing medical radium artifacts. To the public, radium reflected a myth, the powerful, often gendered, myth of the interrelation of science, technology, and medicine. Mass advertisement and popular images of radium enforced that myth. Take for example the following story.
93In Vienna on May 30, 1935, a female journalist from Neues Wiener Journal paid a visit to the Radium Institute to see "seven young Viennese women disintegrate the atom." Although she understands nothing of radium research the journalist has an interest or a "preference" for it, because, as she puts it, radioactivity is a science where every month, there is a new discovery one reads about in the newspaper. Indeed a few months later the Neues Wiener Journal published an article on American efforts to light the streets of the cities and highways using radium.100 The anonymous journalist further argues that radioactivity is "a science that was established by a woman and thus it is romantic. . . .romantic for the amateur."
94In this clear association of romanticism with the presence of women in the field, one can track the public image of radium: a material associated with "the personality of its discoverer, a courageous French lady who was more than a match for a generation of male physicists." Curie's myth coupled with the 1920s myth of radium as a powerful commodity in the service of women, resulted in a positive popular image for the whole discipline. As a romantic endeavor, radioactivity suited women who "remained women despite their work," and they never lost their femininity, as the journalist discovers. Wondering around the institute's laboratories, she comes to a room with the name of a Frau Doctor on its door. The place looks like a witch's kitchen on the stage of Faust. Vessels, glass tubes, and apparatus that are out of her reach were laid around. "On a hook on the wall is hanging an elegant Trotteur-hat and an English, very stylish lady's coat. From this, I see that the Frau Doctor, who disintegrates the atoms and searches for radium in this room, must be a very nice woman; despite the glass tubes and vials, she follows the fashion" the journalist affirms with surprise.101 Similar to the case of Joliot-Curie who had visited the Vienna Institute with her husband a couple of months previously, those women seem to fit a similar model; they are equally women, mothers, partners, and scientists.
95However, as we will witness in the chapters that follow, radium research was far from a romantic endeavor and far different from its public image for most of the women who worked at the Radium Institute, especially toward the end of the 1930s. The point is that radium was never one object, shared among different intersecting worlds. It was always already a different material embedded with different meanings, depending on the hands that held and manipulated it.
^topNotes
Note 1: Curie, M. "Radium and Radioactivity" (1904), 461. back
Note 2: Curie, Pierre. 1905. "Radioactive Substances, Especially Radium" Nobel Lecture, June 6, 1905,
http://nobelprize.org/nobel_prizes/physics/laureates/1903/pierre-curie-lecture.html. back
Note 3: Appadurai, "Introduction: Commodities and the Politics of Value" (1986), 5. back
Note 4: Himstedt, "Radioactivity" (1906). back
Note 5: Hahne, Frank. 1989. "Early Uranium Mining in the United States" in World Nuclear Association: Energy for Sustainable Development, http://www.world-nuclear.org/reference/usumin.html. back
Note 6: I am directly referring here to Leigh Star's and James Griesemer's notion of boundary objects, objects that are shared among several intersecting worlds and have different meanings in each of them, sustaining though a structure common enough to make them recognizable means of translation. (Leigh and Griesemer, "Institutional Ecology, 'Translations,' and Boundary Objects" (1989)). The notion of boundary object, although proved to be extremely useful in several STS studies, does not capture radium's shifting identities between different worlds and the exchange of knowledge and expertise that goes hand in hand with the trade of the material. The focus in the case of the boundary object is on the boundaries that are traversed. The focus here is on the actual trafficking of materials and the culture that supports the traffic. back
Note 7: In her 1911 Nobel lecture, Curie claimed that "Although radium has so far only been obtained in very small amounts, it is nevertheless true to say, in conclusion, that it is a perfectly defined and already well-studied element." The opposite holds for polonium because at the time, scientists were unable to separate it given its low proportion in minerals and its short half life. (Curie, Marie. "Radium and the New concepts in Chemistry" Nobel lecture, December 11, 1911
http://nobelprize.org/nobel_prizes/chemistry/laureates/1911/marie-curie-lecture.html ). back
Note 8: I owe my focus on materials to Ursula Klein and her interesting work on "The Making of Materials," a work in progress which involves a workshop co-organized with Emma Spary at the Max Planck Institute for History of Science in Berlin in 2004 and a forthcoming edited volume, which includes papers by Pamela Smith, Seymor Mauskopf, Augusti Nieto-Galan, Barbara Orland, Ernst Homburg, Christoph Bartels, and Marcus Popplow. My sincere thanks to Klein's generosity for sharing with me the papers of the workshop. back
Note 9: Kohl, "Von den 'Becquerel-Strahlen'" (1997), 489. Dutreix and Dutreix, "Henri Becquerel" (1995) 1870. back
Note 10: Becquerel "Sur les Radiations Invisible" (1896), 501–03. For more on Becquerel's experiments, see: Kohl, "Von den 'Becquerel-Strahlen'" (1997) 488-491; Peh, "The Discovery of Radioactivity" (1996), 627–630. Blaufox, "Becquerel" (1996), 145–154; Dutreix and Dutreix, "Henri Becquerel" (1995), 1869–1875. back
Note 11: Mould, "The Discovery of Radium" (1998); Peh, "The Discovery of Radioactivity" (1996). back
Note 12: Curie, M. "Radium and Radioactivity" (1904), 462. back
Note 13: Mould, "The Discovery of Radium" (1998), 1233; Pycior, "Marie Curie" (1997), 31–50. back
Note 14: Davis, "The Research School of Marie Curie" (1995), 323. back
Note 15: Pycior, "Reaping the Benefits" (1993), 304. back
Note 16: Mazeron and Gerbaulet, "The Centenary of Discovery of Radium" (1998), 207. The original publication of the Curies' discovery appeared in Comptes rendus, "Sur une substance nouvelle radioactive" (1898), 175–78. back
Note 17: Mould, "The Discovery of Radium" (1998), 1233. back
Note 18: Davis, "The Research School of Marie Curie" (1995), 324. back
Note 19: Blaufox, "Becquerel" (1996), 145. back
Note 20: For the reaction of the French chemists, see Peh, "The Discovery of Radioactivity and Radium" (1996), 628. back
Note 21: Curie, M. and P. and Bémont, "Sur une nouvelle substance," (1898). See also Peh, "The Discovery of Radioactivity," (1996). back
Note 22: Mould, "The Discovery of Radium," (1998), 1238. back
Note 23: Pycior, "Reaping the Benefits" (1993), 305; Davis, "The Research School of Marie Curie" (1995), 328. back
Note 24: Törneblach, H. R. Presentation Speech, The Nobel Prize in Physics, December 10, 1903,
http://nobelprize.org/nobel_prizes/physics/laureates/1903/press.html. back
Note 25: Davis, "The Research School of Marie Curie," (1995), 333. back
Note 26: Another chemist and Rutherford's close friend, Bertram Boltwood, centered his work on the chemistry of radioactive elements, assisted later by the Norwegian chemist Ellen Gleditsch. Boltwood was actually the first to obtain a university position in radiochemistry in 1910. Badash, Radioactivity in America, (1979), 15, 64. back
Note 27: The German physicist Friedrich Ernst Dorn first noticed in 1900 that radium was giving off a gas which he named niton, the radium emanation later known as radon. On Brooks, see Rayner-Canham, M. and G. Hariet Brooks: Pioneer Nuclear Scientist (1992). back
Note 28: Shea, Otto Hahn and the Rise of Nuclear Physics (1983). back
Note 29: Feather, Lord Rutherford (1940), 63. Heilborn, Ernest Rutherford and the Explosion of Atom (2003). back
Note 30: Shea, Otto Hahn and the Rise of Nuclear Physic (1983), 5,9. In France, Debierne had discovered actinium and Giesel claimed to have found a different element, emanium. The question was whether the two substances were identical or not. Hahn and Otto Sackur were asked by Ramsay to resolve the controversy. They proved that the substances were actually identical. back
Note 31: On Giesel, see Hahn, "Friedrich Giesel" (1928). Especially on Mendeleev's work on radioactivity, see Gordin, A Well-Ordered Thing (2004). back
Note 32: Feather, Lord Rutherford (1940), 105. back
Note 33: Badash, Radioactivity in America (1979), 205. On Ramsay and the radium industry in England see also Harvie, "The Radium Century" (1999). back
Note 34: Gordin, A Well-Ordered Thing (2004). back
Note 35: Nye, The Question of the Atom (1986), 456. back
Note 36: Badash, Radioactivity in America (1979), 69. back
Note 37: Nye, The Question of the Atom (1986), 430; Gordin, A Well-Ordered Thing (2004), 226. back
Note 38: Curie, Pierre. "Radioactive Substances, Especially Radium" Nobel lecture, June 6, 1905,
http://nobelprize.org/nobel_prizes/physics/laureates/1903/pierre-curie-lecture.html;
Curie, Marie. "Radium and the New Concepts in Chemistry," Nobel lecture, December 11, 1911,
http://nobelprize.org/nobel_prizes/chemistry/laureates/1911/marie-curie-lecture.html. back
Note 39: Kaiserliche Academie der Wissenschaften in Wien, Konzepte, no. 20 015, AÖAW. back
Note 40: The Curies to the Austrian Academy, 1898, no. 25 081, AÖAW. back
Note 41: Karlik and Schmid, Franz Serafin Exner, (1982), 85; Fischer, Geschichte der Gesellschaft der Ärzte (1938), 155. back
Note 42: Julian, "The Early Days of the X-Ray Revolution" (1996), 39. See also Lesky, The Vienna Medical School of the 19th Century (1976), 304. back
Note 43: Fischer, Geschichte der Gesellschaft der Ärzte, (1938), 155. Karlik and Schmid, Franz Serafin Exner (1982), 86; Alth, 50 Jahre Strahlentherapie Lainz (1981), 4; Kogelnik, "The History and Evolution of Radiotherapy" (1996), 221. back
Note 44: Pierre Curie received Suess's telegram via Michel Levy. Between 1898 and 1899, the Curies received 1.1 tons of pitchblende from the Austrians. By 1906, the amount went up to 23.6 tons. The first amount was supplied at no cost and the ones followed were sold for especially low prices. See Kaiserliche Academie der Wissenschaften in Wien, Konzepte, no. 20 015, AÖAW; Pierre Curie to Suess, November 17, 1898, AÖAW; Meyer, "Die Vorgeschichte" (1950), 1; Reiter, "Stefan Meyer" (2001), 110. back
Note 45: Curie, M. "Radium and Radioactivity" (1904), 462; Curie, Marie "Radium and the New Concepts in Chemistry" Nobel lecture, December 11, 1911, http://nobelprize.org/nobel_prizes/chemistry/laureates/1911/marie-curie-lecture.html; Roquè, "Marie Curie and the Radium Industry" (1997), 270. back
Note 46: See for example the case of Ellen Gleditsch. In 1916, Curie prompted her to come to France and work at the factory, hoping that "He will offer you good conditions." Gleditsch followed Curie's advice and took Debierne's position at the factory while he was serving at the front. Sharing her time between the factory and Curie's laboratory in several other occasions, Gleditsch was able to pursue her research. (Lykknes, Ellen Gleditsch: Professor, Radiochemist, and Mentor (2005), 134). back
Note 47: "Seeks Radium in Colorado" The New York Times, s12, 1:6, September 12, 1912. back
Note 48: Roquè, "Marie Curie and the Radium Industry" (1997), 268. back
Note 49: Reiter, "Stefan Meyer" (2001), 109–10; Meyer, "Die Vorgeschichte" (1950), 1; Meyer, "Magnetic Properties of the Elements," (1899); Meyer, "Magnetic Properties of Inorganic Compounds" (1899). Meyer had studied physics and chemistry at the University of Vienna and was able to combine both in his early research on radioactivity. back
Note 50: Suess and Lang to Auer von Welsbach, July 10, 1901, AÖAW. back
Note 51: Meyer, "Die Vorgeschichte" (1950), 10. back
Note 52: The French, for example, carried out the purification of the raw material in the Central Company for Chemistry Production (Société Central de Produits Chimiques) a spacious factory where the chemist André Debierne could handle the enormous amounts of pitchblende. Only after the first extraction was further work completed at Curie's laboratory. As Soraya Boudia points out, Debierne "had to adapt laboratory techniques and perfect industrial treatment methods." The company provided chemical products and the staff salaries. In return, they obtained a share of the extracted radium salts for marketing. The forced collaboration of chemists and physicists with industrialists, as Boudia emphasizes, played an important role to the construction of the radioelement industry in France. (Boudia, "The Curie Laboratory" (1997), 250–51; Mazeron and Gerbaulet, "The Centenary of Discovery" (1998), 207). back
Note 53: For Auer von Welsbach scientific discoveries, see Auer von Welsbach Museum,
http://www.althofen.at/AvW-Museum/Englisch/biographie_e.htm. Also "Carl von Welsbach, the Inventor of the Incandescent Light Mantle," (1912). back
Note 54: "Haitinger-Feier," Chemiker-Zeitung, (1930), 182. back
Note 55: Suess to Auer von Welsbach, July 10, 1901, AÖAW. back
Note 56: Reiter, "Stefan Meyer" (2001), 111. back
Note 57: Up to that time, the mines in St. Joachimsthal were the main and only considerable source of pitchblende in the world. Echoing Boltwood, Lawrence Badash argues that, the embargo reflects Austrians' displeasure with the commercial instead of the mere scientific use of radium. Badash, Radiaoctivity in America, (1979), 140. back
Note 58: Suess and Lang to the Ministry of Agriculture, January 15, 1904, AÖAW; Suess and Lang to the Ministry of Culture and Education, January 15, 1904, AÖAW. back
Note 59: Suess and Lang to the Ministry of Agriculture, January 15, 1904, AÖAW. back
Note 60: On March 7, 1904 Suess and Lang informed Pierre Curie of their agreement with the Ministry of Agriculture. Given the shortage of the residue, they agreed that the two research groups, the Austrians and French, would alternate receiving one ton each at a time On March 22, Lang and Suess informed the administrators of the St. Joachimsthal mines of the above procedure. The mine's administrators would send alternatively one ton of pitchblende to Atzgersdorf, Vienna and one to Paris. Further financial issues were clarified. Between 1904 and 1905, the Curies paid 9,540 kronen to the mines for the residues. See Suess and Lang to Pierre Curie, March 7, 1904, AÖAW; Suess and Lang to k.k. Berg- und Hüttenverwaltung, St. Joachimsthal, March 22, 1904, AÖAW; Meyer, "Die Vorgeschichte" (1950), 9. back
Note 61: Haitinger and Ulrich, "Berich über die Verarbeitung," (1908); "Haitinger-Feier" Chemiker-Zeitung, (1930), 182; Meyer, "Das erste Jahrzehnt," (1920), 1; Exner, "Le nouvel institute" (1910), 244. back
Note 62: The Royal Society to the Austrian Academy, May 1, 1904, AÖAW. back
Note 63: Rona, How it Came About (1978), 21. back
Note 64: Cahan, An Institute for an Empire (1989), 8; Warburg to Exner, February 12, 1909, AÖAW; Kaiserliche Academie der Wissenschaften in Wien, Konzepte, 25 015, AÖAW. For example, a list of radium recipients included the Chemistry Institute in Krakau and Eduard Riecke's institute at the University of Göttingen (1908 report of the Ministry of Public Affairs, n. 904, AÖAW). back
Note 65: Meyer, "Die Vorgeschichte" (1950), 14. back
Note 66: For characterizations of Austria as the center of the monarchy's power, see Okey, The Habsburg Monarchy (2001), 196. When the Austro-Hungarian Empire collapsed, Ulrich lost his position as director at the St. Joachimsthal mines and moved to Vienna as a guest at the Institute for Radium Research (Meyer, "Das erste Jahrzehnt" (1920), 11). back
Note 67: Suess and Lang to the Ministry of Culture and Education, March 14, 1908, AÖAW. back
Note 68: Walkhoff published a brief description of a skin reaction to radium in October 1900. Interestingly enough, he published his report in a journal of amateur photography. This fact calls attention to the interrelation of physics, medicine, and photography. (Walkhoff, "Unsichtbare, photographische wirksame," (1900). See also Dutreix, Tubiana and Pierquin, "The Hazy Dawn of Brachytherapy," (1998), 223.); Bequerel and Curie, "Action physiologiques," (1901). As Mould correctly emphasizes, Pierre Curie and Bequerel planned their experiments and they were not accidental (Mould, "The Discovery of Radium" (1998), 1241). Radium had already started to occupy the interest not only of physicists and physicians but also of the general public. For example, in 1903, the American inventor Thomas Edison warned the public of radium's possible damaging biological effects in an interview in Penny Press (see Mullner, Deadly Glow (1999), 16). back
Note 69: Tubiana, Dutreix, and Pierquin, "One Century of Radiotherapy" (1996), 231; See also Viol, "History and Development of Radium Therapy" (1921), 21. back
Note 70: Hayter, An Element of Hope (2005), 13. back
Note 71: On the radium therapy in France see Vincente, "Genesis of the Pavillion Pasteur of the Institut du radium in Paris"(1997). back
Note 72: Berven, et al., The First Fifty Years (1965). back
Note 73: Fox, "The History of Radium" (1998), 115–124. back
Note 74: Hayter, An Element of Hope (2005), 13; Sell, "The Development of Radiotherapy in Denmark During 100 Years" (1995); Alth, 50 Jahre Strahlentherapie Lainz (1981), 4–5; Luger, 70 Jahre Krankenhaus (1977), 3; Mould, A Century of x-rays (1993), 136. back
Note 75: Exner, S. "Einige Beobachtungen" (1903); Exner, A. and Holzknecht, " Die Pathologie" (1903). Alfred Exner was the first to treat a tumor of the esophagus by forcing a catheter that held the radioactive source through the esophagus so as to achieve improvement in swallowing (Exner, A. "Über die Behandlung," (1904), 4–96). See also Fischer, Geschichte der Gesellschaft der Ärzte, (1938), 156. In 1902, Antoine Beclère, an outstanding pioneer of radiology in France, visited Holzknecht in Vienna to discuss dosimetry issues. In 1905, Holzknecht took over the leadership of the Röntgen Laboratory of the second Medical University Clinic of Vienna, later known as the Holzknecht Institute, elevating radiology to an exact science and Vienna to the center of radiological research. Five years later, Holzknecht joined Freud's psychoanalytical society after experiencing psychological anxieties. More than two decades later, in 1931, Holzknecht treated Freud's tumor in his institute. On Holzknecht see Angetter, Guido Holzknech, (1998), 16–17; Tubiana, Dutreix, and Pierquin, "One Century of Radiotherapy" (1996), 228. back
Note 76: Tubiana, Dutreix, and Pierquin, "One Century of Radiotherapy" (1960), 231. Also, for illustrations of early radium therapy equipment and methods, see Mould, A Century of x-rays, (1993), 27, 131. See also http://www.orau.org/ptp/collection/brachytherapy/brachytherapy.htm. back
Note 77: Report of the Ministry of Public Affairs drafted in 1908 n. 904, AÖAW. In 1904, Suess was interviewed by Louis Leasley from London. On his return to London, Leasley asked Suess for a radium loan in order to pursue "hospital work" (Leasley to Suess, February 18, 1904, no. 25 083, AÖAW). back
Note 78: Rutherford to Meyer, June 1, 1914, AÖAW; Rutherford to Meyer, June 29, 1914, AÖAW. back
Note 79: DiSantis, "Radiologic History Exhibit" (1991). For more on advertisements concerning racial issues, see Massing, "From Greek Proverb to Soap Advert" (1995). back
Note 80: Wiesloch, 1916. "Sterilization in the Interest of Race Betterment" (1916). back
Note 81: Chase, "American Literature on Radium and Radium Therapy prior to 1906" (1921); Viol, "History and Development of Radium Therapy" (1921); Soddy, "Radium as a Therapeutic Agent" Medical Record" (1903). back
Note 82: Ernest Rutherford to Stefan Meyer, October 22, 1910, Rutherford's Papers, Manuscript Collections, CUL. back
Note 83: "Urges that Radium Be Nationalized" New York Times, November 30, 1913, III, 4:1. back
Note 84: Mullner, Deadly Glow (1999), 21–29; The Historical Instrumentation Collection, Oak Ridge Associated Universities,
http://www.orau.org/ptp/collection/Miscellaneous/photoalbum/photoalbum.htm back
Note 85: Boudia, "Radioactivity and Metrology" (1997), 251. back
Note 86: Mache, "Über die im Gasteiner Wasser" (1904a); Mache, "Über die im Gasteiner Wasser," (1904b); Mache and Meyer, "Über die Radioaktivität der Quellen" (1905). back
Note 87: Mould, A Century of x-rays (1993), 170. back
Note 88: "Controls its Own Radium" The New York Times, November 30, 1911, N22, 22:2; Meyer, "Die Vorgeschichte" (1950), 3. back
Note 89: "Re Präparate," newspaper clip, undated, AÖAW. back
Note 90: Meyer, "Die Vorgeschichte" (1950), 11. back
Note 91: For radioactive products, see The Historical Instrumentation Collection, Oak Ridge Associated Universities, http://www.orau.org/ptp/collection/quackcures/quackcures.htm; Radiation and the Popular Imagination, Radiology Centennial Inc., formerly available at http://www.xray.hmc.psu.edu/rci/contents_8.html and archived at http://web.archive.org/web/20060504024805/http://www.xray.hmc.psu.edu/rci/contents_8.html (accessed 7 November 2007). See also Baker, "Industrial Uses of Radium: Possibilities in Agriculture, Medicine, and Other Directions" (1915). back
Note 92: For most of the advertisements and products presented here, see http://www.orau.org/ptp/museumdirectory.htm. back
Note 93: "The Radiendocrinator"
http://www.orau.org/ptp/collection/quackcures/radend.htm; Mullner, Deadly Glow (1999), 109–118. back
Note 94: Photograph VIII-09, "Beautifying x-rays", Radiation and the Popular Imagination, Radiology Centennial Inc. Formerly available at http://www.xray.hmc.psu.edu/rci/ss8/ss8_1.html; archived at http://web.archive.org/web/20051125035151/http://www.xray.hmc.psu.edu/rci/ss8/ss8_1.html (accessed 7 November 2007). back
Note 95: "Radium and Beauty," New York Tribune, November 10, 1918, Magazine Section, p. 12; Photograph VIII-10, "Beauty Shop Horrors" Radiation and the Popular Imagination, Radiology Centennial Inc., formerly available at http://www.xray.hmc.psu.edu/rci/ss8/ss8_1.html and archived at http://web.archive.org/web/20051125035151/http://www.xray.hmc.psu.edu/rci/ss8/ss8_1.html (accessed 7 November 2007). back
Note 96: As Soraya Boudia argues, it is not entirely certain that Alfred Curie actually existed. The authors of the article draw attention to the fact that Alfred's last name signified scientific authority and thus those involved in the commercialization of Tho-Radia probably aimed to secure commercial success for the new product. See Lefebvre, and Raynal, "De l' Institut Pasteur à Radio Luxemburg: L' histoire étonnante du Tho-Radia" (2002); "la curieuse histoire du Tho-Radia" http://monsite.wanadoo.fr/tho-radia/index.jhtml. For a history of women's beauty trend, see Riordan, Inventing Beauty (2004). back
Note 97: Riordan, Inventing Beauty (2004). See also
http://www.double-d.com/coffeebreak/companies/silhouette.html and http://homepage.ntlworld.com/davesplace/strange.htm. back
Note 98: Lang, "A Most Valuable Accident" (1959). See also Clark, Radium Girls (1997). back
Note 99: Rentetzi, "Women Radium Dial Painters as Experimental Subjects or What Counts as Human Experimentation" (2004). back
Note 100: "Strassenbeleuchtung durch Radium" Neues Wiener Journal, November 10, 1935. back
Note 101: "Sieben junge Winnerinnen zertrümmern Atome…" Neues Wiener Journal, May 30, 1935. I owe my thanks to Brigitte Bischof who brought this article to may attention. Bischof, "The 'Marie Curie' Syndrome" (2003). back
^top











